Call Now: +91 40 2988 7441
Call Now: +91 40 2988 7441
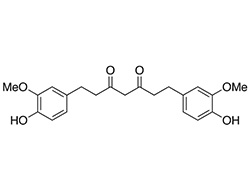
Molecular Weight: 372.41
Formula: C21H24O6
CAS Number: 36062-04-1
HYDROCURCUMINS
Hydrocurcumins, a colorless hydrogenated product derived from the yellow curcumins, (the biologically active principles from the rhizomes of Curcuma longa), function as efficient antioxidant compounds. Hydrocurcumins exhibit many of the same physiologic and pharmacological activities as curcumins and in some systems may exert greater antioxidant activity than curcumins.The superior antioxidant property of Hydrocurcumins, combined with the lack of yellow color, render this product useful in achromatic food and cosmetic applications that currently employ conventional synthetic antioxidants .
Source
Hydrocurcuminoids are derived from curcuminoids (extracted from the roots of Curcuma longa, commonly called turmeric rhizomes).
Potential Uses
Dietary Supplements
Nutritional Products
Cosmetics
Antioxidant
One study evaluated the comparative antioxidant activity of curcuminoids and TetraHydrocurcumins invitro using linoleic acid as the substrate in an ethanol/ water system as well as using rabbit erythrocyte membrane and rat liver. It was found that TetraHydrocurcumin had the strongest antioxidant activity among all curcuminoids in each assay system. The authors concluded that these results suggest that TetraHydrocurcumin must play an important role in the antioxidant mechanism of Curcumin in vivo .
Curcumin and four synthetic analogs were examined for anti-inflammatory potential in carrageenin induced foot paw edema and cotton pellet granuloma models of inflammation in rats , . Hydrocurcumin was effective at half the dose of the parent compound, curcumin. Comparison of curcumin and its analogs in acute and sub acute models of inflammation revealed that curcumin analogs are more active in alleviating acute inflammation
In one of the studies significant inhibition of ACF (aberrant crypt foci) development in the colons of mice treated with fucoxanthin, lutein or Hydrocurcumins when given in the post-initiation phase (tumors were initiated using DMH) was observed. The dose dependent decreases of BrdU LI observed for lycopene and Hydrocurcumin indicate that larger doses might be more effective for inhibition of ACF development. This study demonstrated that Hydrocurcumin is more active than the parent compound, curcumin, in terms of inhibition of ACF development and cell proliferation. This observation combined with the fact that Hydrocurcumin which has both phenolic and beta-diketone moieties in the same molecule, is a stronger antioxidant suggests that Hydrocurcumin might be particularly suitable for application as a chemo preventive agent against in vivo carcinogenesis.
Free radical chain reactions are implicated in most degenerative biological reactions. Free radicals on the surface of the skin, generated through exposure to ultraviolet radiation, chemicals or other environmental stress factors catalyze aging of the skin. Hydrocurcumin scavenges free radicals, thus preventing their formation. Additionally, these compounds also inhibit the propagation of free radicals. These antioxidants also help to improve the shelf life of fat-based topical formulations by inhibiting the autoxidation of fats. The anti-inflammatory effects of Hydrocurcumin combined with their efficient antioxidant action render them useful as ingredients in anti-aging formulations and in topical formulations designed to maintain general skin health and integrity.
* Note: Intended use based on published data and traditional practices. Therefore No Warranty is expressed or implied
PRODUCT SPECIFICATION
| QUALITY CONTROL DEPARTMENT | |
| Product Name | White Curcumin |
| Botanical Name | Curcuma longa |
| Plant part | Rhizomes |
| Molecular formula | Hydrocurcumins |
| S. No. | Parameters | Specification | Method | ||||||||||||||||||
| Physical | |||||||||||||||||||||
| 1 | Description | White to pale yellow colored powder | Visual | ||||||||||||||||||
| 2 | Identification | To comply by HPLC | KVN/QC/STP-IN/030 | ||||||||||||||||||
| 3 | Loss on drying | NMT 5.0% | USP <731> | ||||||||||||||||||
| 4 | Residue on Ignition | NMT 6.0% | USP <281> | ||||||||||||||||||
| 5 | Tapped bulk density | 0.30 -0.80g/ml | USP <616> | ||||||||||||||||||
| Sieve test (passes through) | |||||||||||||||||||||
| 6 |
|
||||||||||||||||||||
| Sieve test (passes through) | |||||||||||||||||||||
| Chemical | |||||||||||||||||||||
| 7 | Purity by HPLC Sum of all Hydrocurcumins | NLT 99.0% w/w on dry basis | KVN/QC/STP-IN/030 | ||||||||||||||||||
| 8 | Assay by HPLC Sum of all Hydrocurcumins | NLT 95.0% w/w on dry basis | KVN/QC/STP-IN/030 | ||||||||||||||||||
| Others | |||||||||||||||||||||
| 9 |
|
||||||||||||||||||||
| 10 | Residual solvents | Should comply by ICH/USP | USP <467> | ||||||||||||||||||
| Microbiological Profile | |||||||||||||||||||||
| 11 |
|
||||||||||||||||||||
We will provide the Spectral Data on Request