Call Now: +91 40 2988 7441
Call Now: +91 40 2988 7441
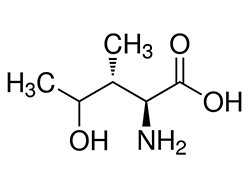
NAME: 4-Hydroxyisoleucine [Trigonella foenum-graecum seed extract]
4-Hydroxyisoleucine is a non protein amino acid, isolated from Fenugreek seeds (Trigonella foenum-graecum). Fenugreek has a long history of use in the traditional Indian (Ayurveda) and Chinese medicines. It is a potential product as anti-diabetic, anti-dyslipidemic, muscle building, athletic diet and for skin and scalp health care applications.
4-Hydroxyisoleucine
Molecular Formula: C6H13NO3
Molecular weight: 147.17232 g/mol
Anti – diabetic activity:
4-Hydroxyisoleucine was demonstrated to stimulate glucose – dependent insulin by direct effect on pancreatic islets (Sauvaire et.al.1991).The insulin stimulating properties of 4-Hydroxyisoleucine are quite different from those of sulfonylureas. It acts on the pancreatic cells and stimulate the insulin secretion only when the glucose secretion increases above the norm. (Hyperglycemia) and also improves insulin sensitivity. It is a potential therapeutic agent for diabetics (Broca et.al, 2004).
Anti – dyslypidemic activity:
4-Hydroxyisoleucine has also shown significant anti-dyslipidemic activity by decreasing the levels of plasma triglycerides, total cholesterol and free fatty acids in a dyslipidemic hamster model (Narender et.al, 2006).
Muscle building:
Branched chain amino acids, isoleucine, leucine and valine are important to muscle growth and 4-Hydroxyisoleucine, being a branched chain amino acid, is likely to play a similar role. The combination of 4-Hydroxyisoleucine with other amino acids could exert unique physiological responses such as increase in gut absorption of glucose, stimulation of pancreatic beta cells, and enhanced disposal of other carbohydrates (Lee et.al, 2010). 4-Hydroxyisoleucine has a nutrient partitioning effect, which means it may help to shuttle nutrients to muscle cells preferentially over fat cells.
PRODUCT SPECIFICATION
| QUALITY CONTROL DEPARTMENT | |
| Product Name | 4-Hydroxyisoleucine 40% |
| Botanical Name | Trigonella foenum-graecum |
| Plant part | Seeds |
| Standardization | 4-HIL [4-Hydroxyisoleucine] |
| S. No. | Parameters | Specification | Method | ||||||||||||||||||
| Physical | |||||||||||||||||||||
| 1 | Description | Off white to gray to Brown colored | Visual | ||||||||||||||||||
| 2 | Identification | To comply by HPLC | KVN/QC/STP-IN/029 | ||||||||||||||||||
| 3 | Loss on drying | NMT 5.0% | USP <731> | ||||||||||||||||||
| 4 | Residue on Ignition | NMT 6.0% | USP <281> | ||||||||||||||||||
| 5 | Tapped bulk density | 0.30 -0.80g/ml | USP <616> | ||||||||||||||||||
| Sieve test (passes through) | |||||||||||||||||||||
| 6 |
|
USP <786> | |||||||||||||||||||
| Chemical | |||||||||||||||||||||
| 7 | Assay by HPLC 4-Hydroxyisoleucine |
NLT 40.0% w/w on dry basis |
KVN/QC/STP-IN/029 |
||||||||||||||||||
| Others | |||||||||||||||||||||
| 8 |
|
||||||||||||||||||||
| 9 | Residual solvents | Should comply by ICH/USP | USP <467> | ||||||||||||||||||
| Microbiological Profile | |||||||||||||||||||||
| 10 |
|
||||||||||||||||||||